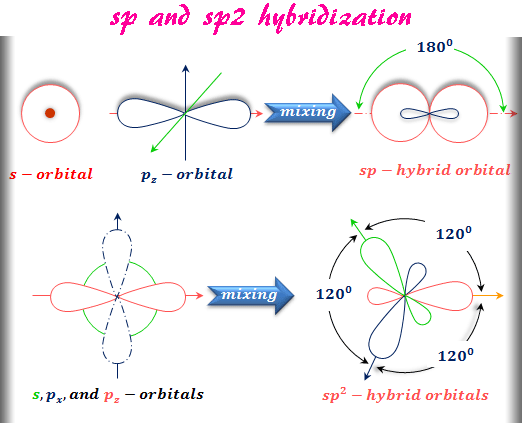
Each sp hybrid orbitals has 50% s-character and 50% p-character. Sp Hybridisation : This type of hybridization involves the mixing of one s and one p orbital resulting in the formation of two equivalent sp hybrid orbitals. (iv) Completely filled orbitals of valence shell can also take part in hybridization. (iii) Promotion of electrons is not an essential condition before hybridization. (ii) The orbitals undergoing hybridization should have almost equal energy. (i) The orbitals present in the valence shell of the atom are hybridized. (iv) These hybrid orbitals are directed in space in some preferred direction to have minimum repulsion between electron pairs and thus a stable arrangement. 
(iii) The hybrid orbitals are more effective in forming stable bonds than the pure atomic orbitals. 
(ii) The hybridized orbitals are always equivalent in energy and shape. (i) The number of hybrid orbitals is equal to the number of atomic orbitals that get hybridized. Hybridization : It can be defined as the process of intermixing of the orbitals of slightly different energies so as to redistribute their energies, resulting in the formation of a new set of orbitals of equivalent energies and shape.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
